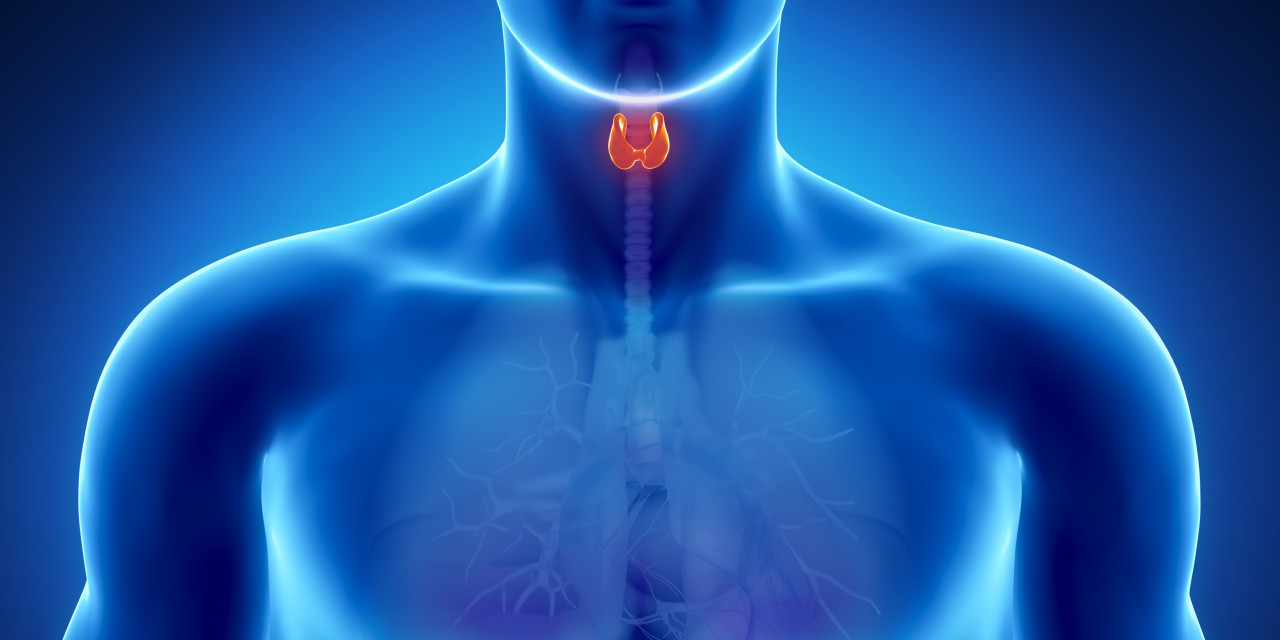
Hypothyroidism, otherwise known as ‘underactive thyroid’ is one of the most common endocrine diseases, second only to diabetes.1 Symptoms vary widely and include tiredness, weight gain, dry skin, intolerance to cold, constipation, muscle weakness, impaired memory, poor concentration, depression, infertility and even hair loss of the outer third of eyebrows.2, 3 The thyroid acts as both the accelerator and brake pedal in the body and regulates energy and heat production, as well as normal growth, tissue repair and cell development.1
When investigating thyroid dysfunction, it is important to think of the endocrine system as whole, because the production and regulation of thyroid hormones are controlled by the same parts of the brain that manage hormones related to the reproductive system, adrenal health, cell growth and kidney function. A part of the brain called the hypothalamus works behind the scenes, acting as an interface between the body and its environment, for example responding to temperature or stress. The hypothalamus sends signals to the pituitary gland at the base of the brain, telling it to pass on instructions to specific organs or cells in the body, including the thyroid gland, to either stimulate or inhibit the release of hormones. Although the pituitary gland is only the size of a pea, it is also called the ‘master gland’ because it is at the centre of this chain of communication.
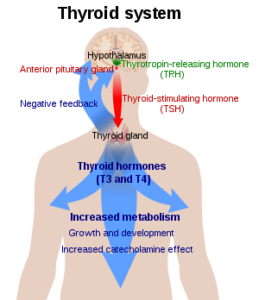
Hormones are essentially chemical messengers and the above diagram shows the main hormones involved in thyroid function. The hypothalamus produces thyrotropin releasing hormone (TRH), which instructs the pituitary gland to release thyroid stimulating hormone (TSH). TSH travels in the blood to its target organ, the thyroid gland, where it binds to receptors on the outside of the thyroid and causes it to secrete thyroid hormones.
‘…from a large pool of thyroid hormones, only a small number of them do the bulk of the work.’
Thyroid hormones are made from the amino acid tyrosine, joined to atoms of iodine. They vary according to the number of iodine atoms they are joined to, with the majority of thyroid hormones having 4, hence the name T4, also known as thyroxine. Molecules with 3 atoms of iodine are referred to as T3 and some T4 is converted to T3 in the liver and kidneys.4 Thyroid hormones are poorly soluble in water and almost 99% of those which are circulating in the blood are attached to carrier proteins.4 The small proportion which are not bound to proteins are called free T3 or free T4 and it is free T3 which plays the most active role in influencing body cells. In other words, from a large pool of thyroid hormones, only a small number of them do the bulk of the work.4
To illustrate how this all works together, imagine a cold winter’s day. Your hypothalamus detects the drop in temperature and initiates the process through which your body can burn fat or glucose to produce energy and heat. The hypothalamus sends a signal (TRH) to the pituitary gland, which then releases TSH into the blood, to pass on the message to the thyroid gland to secrete T4. T4 is converted to T3, the active form of thyroid hormone, which is carried around the body by a carrier protein, then detaches and becomes Free T3, capable of instructing all cells to produce energy. The process of energy production also releases heat, causing body temperature to rise, so you warm up!
A classic symptom of an underactive thyroid is sensitivity to cold, in particular, cold hands and feet.2 A person with an underactive thyroid gland might be receiving signals from the hypothalamus and pituitary to produce thyroid hormones, i.e. they might have high levels of TSH, but there may be some issue further down the chain, whereby the thyroid does not or cannot secrete enough T4. Even if adequate levels of T4 are being secreted and are available in the blood, the issue could be that this is not being converted to its active form of T3 and so it is very important to test T3 levels, not just T4. Furthermore, there is another form of T3 called reverse T3 (rT3), which generally serves as the body’s brake pedal, slowing down metabolic processes at the cellular level and subsequently slowing down brain function, heart rate and the rate at which food is converted to energy.5, 6 Therefore, we also need to measure rT3, in case excess levels are causing weight gain, low energy, loss of libido, poor concentration and other symptoms.
So why might this communication breakdown occur? There are many potential factors involved in primary thyroid failure, including genetics, illness and surgery, such as tonsillectomy which is performed very close the thyroid gland. Pregnancy can affect thyroid hormones, particularly post-partum and certain medications such as lithium or high doses of corticosteroids can influence the chain of communication.7, 8 In addition, environmental toxins such as chlorine or fluoride, or heavy metals such as mercury and lead can also have a disruptive effect, by blocking the uptake of thyroid hormones into cells.7,9
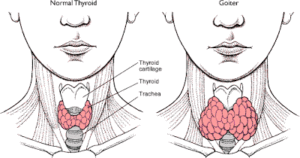
Auto-immune conditions such as Hashimoto’s thyroiditis are the most common cause of hypothyroidism.1,2,3 Immune cells attack the thyroid gland, causing it to become inflamed and enlarged, sometimes resulting in a visible swelling, or goitre at the front of the neck. Over time, damage to the thyroid gland can reduce production of thyroid hormones, causing hypothyroidism, but for many people, they are unaware of symptoms for years. When the immune cells attack the thyroid, antibodies are produced and laboratory tests can check for the presence of these antibodies, to help ascertain the cause of hypothyroidism. It is not always routine practice to check for anti-thyroid antibodies and yet, someone with normal levels of TSH and T4 may be suffering from Hashimoto’s thyroiditis, so it is well worth requesting this test at the same time as your thyroid hormone test.
From a nutritional perspective, deficiencies in the nutrients required for the production and uptake of thyroid hormones can affect hormone levels. Essential fats are required for the uptake of thyroid hormones by cells.4 Tyrosine and iodine are both needed for production of T4, along with vitamins A and C, B vitamins, copper and zinc.4 Iodine is depleted in the soil of many countries, including the UK, Australia and NZ10 and iodine-related goitre is more prevalent in women than men.11 Certain foods are labelled as goitrogenic because they block utilisation of iodine by the body.2 They include raw brassicas such as broccoli, cabbage, kale, cauliflower and Brussels sprouts, plus soya beans, millet and pine nuts.9 At $88 per kilo, I’d advise anyone to go easy on the pine nuts, but don’t be put off eating your greens, as lightly cooking them renders their goitrogenic properties inactive2 and you would have to eat a large quantity of raw brassicas to experience an effect!
‘reverse T3 generally serves as the body’s brake pedal, slowing down metabolic processes at the cellular level and subsequently slowing down brain function, heart rate and the rate at which food is converted to energy.’
 The conversion process of T4 to T3 in the liver and kidneys also requires copper, zinc and magnesium and may be inhibited by selenium deficiency2, 9 (selenium is also deficient in NZ soil and the NZ population),12 or excess levels of iodine.9 The liver and kidneys need to be functioning well in order to effectuate the conversion, so any compromise, particularly to liver health can be detrimental and is something to consider if you have experienced any digestive problems, which have a knock on effect on the liver.4 Similarly, if adrenal function is suboptimal and levels of the stress hormone cortisol are low, this can have a significant effect on the balance of thyroid hormones, resulting in high levels of TSH, as the brain tries to stimulate production, but T4 production by the thyroid gland is reduced and a greater proportion of whatever T4 is produced, is converted to rT3, the inactive form of T3, which slows down the body’s metabolic processes. In addition, low levels of cortisol, which are often associated with chronic stress and adrenal insufficiency13 may further inhibit the conversion of T4 to active T3 in the liver and kidneys.
The conversion process of T4 to T3 in the liver and kidneys also requires copper, zinc and magnesium and may be inhibited by selenium deficiency2, 9 (selenium is also deficient in NZ soil and the NZ population),12 or excess levels of iodine.9 The liver and kidneys need to be functioning well in order to effectuate the conversion, so any compromise, particularly to liver health can be detrimental and is something to consider if you have experienced any digestive problems, which have a knock on effect on the liver.4 Similarly, if adrenal function is suboptimal and levels of the stress hormone cortisol are low, this can have a significant effect on the balance of thyroid hormones, resulting in high levels of TSH, as the brain tries to stimulate production, but T4 production by the thyroid gland is reduced and a greater proportion of whatever T4 is produced, is converted to rT3, the inactive form of T3, which slows down the body’s metabolic processes. In addition, low levels of cortisol, which are often associated with chronic stress and adrenal insufficiency13 may further inhibit the conversion of T4 to active T3 in the liver and kidneys.
At the top of the chain, there is the possibility that damage to the hypothalamus or pituitary, caused by a tumour, radiation or infection for example, can result in insufficient levels of TSH being produced, which slows down the whole process.
 As you can see, there is a lot to consider and it is important to identify where the breakdown in communication has occurred, rather than just fixing the problem by medicating with a synthetic version of thyroid hormone, which is the standard protocol. Although hypothyroidism is relatively easy to treat from a medical perspective, many studies highlight problems with management of the condition, with under or over-replacement of thyroid hormone each creating its own issues.3,7 Another major issue is that levothyroxine, the most commonly prescribed synthetic thyroid replacement hormone, provides T4 only. If a patient is unable to convert T4 to T3, then this may not solve the problem and while both natural and synthetic forms of T3 medication are widely available in the US, they are not generally approved or funded in the UK or NZ, which leaves many sufferers feeling no better while taking levothyroxine3, 14 and forces them to source their own alternatives.
As you can see, there is a lot to consider and it is important to identify where the breakdown in communication has occurred, rather than just fixing the problem by medicating with a synthetic version of thyroid hormone, which is the standard protocol. Although hypothyroidism is relatively easy to treat from a medical perspective, many studies highlight problems with management of the condition, with under or over-replacement of thyroid hormone each creating its own issues.3,7 Another major issue is that levothyroxine, the most commonly prescribed synthetic thyroid replacement hormone, provides T4 only. If a patient is unable to convert T4 to T3, then this may not solve the problem and while both natural and synthetic forms of T3 medication are widely available in the US, they are not generally approved or funded in the UK or NZ, which leaves many sufferers feeling no better while taking levothyroxine3, 14 and forces them to source their own alternatives.
“Many patients who present with symptoms of thyroid dysfunction return normal test results.”
 A vital tool for a correct diagnosis is a comprehensive test, which looks not just at TSH and T4, but also measures free T3, rT3 and also anti-thyroid antibodies. Many patients who present with symptoms of thyroid dysfunction return normal test results, but in too many cases, budget does not allow for the full range of analytes to be tested and what is called ‘subclinical’ hypothyroidism remains undiagnosed. There are two important points to note here. Firstly, there is much debate over what constitutes a ‘normal’ thyroid hormone range, given that hormone profiles vary by age, sex and ethnicity, at different trimesters of pregnancy and even at different times of day3, 10, 15. The current accepted reference range for TSH in the UK and NZ is 0.4 to 4.0, while studies have indicated that 95% of healthy subjects have a TSH level below 2.5 and many scientists call for the reference range to be lowered.16 At the very least, it would be prudent to analyse results, guided by an ‘optimal’ reference range, which might highlight potential thyroid dysfunction in less extreme cases.
A vital tool for a correct diagnosis is a comprehensive test, which looks not just at TSH and T4, but also measures free T3, rT3 and also anti-thyroid antibodies. Many patients who present with symptoms of thyroid dysfunction return normal test results, but in too many cases, budget does not allow for the full range of analytes to be tested and what is called ‘subclinical’ hypothyroidism remains undiagnosed. There are two important points to note here. Firstly, there is much debate over what constitutes a ‘normal’ thyroid hormone range, given that hormone profiles vary by age, sex and ethnicity, at different trimesters of pregnancy and even at different times of day3, 10, 15. The current accepted reference range for TSH in the UK and NZ is 0.4 to 4.0, while studies have indicated that 95% of healthy subjects have a TSH level below 2.5 and many scientists call for the reference range to be lowered.16 At the very least, it would be prudent to analyse results, guided by an ‘optimal’ reference range, which might highlight potential thyroid dysfunction in less extreme cases.
The second point which I tend to find with nutrition clients is that it is essential to analyse test results in conjunction with symptoms. Time and time again, individuals receive ‘normal’ test results, yet their symptoms strongly indicate that there is an issue with their thyroid gland. Again, this relates to the definition of ‘normal’ and while one person with levels of TSH at 3.8 may feel in optimal health, many others might continue to struggle with weight gain, fatigue, poor skin, or many of the numerous symptoms associated with an underactive thyroid. Thyroid dysfunction is common among women over the age of fifty17 and many cases of subclinical hypothyroidism remain undiagnosed post-menopause, which is understandable given the broad range and overlap of symptoms associated with this period of hormonal change. However, again it is important that testing is thorough and combined with analysis of symptoms, in order to achieve the correct diagnosis and medical or nutritional protocol.
 In order to support your thyroid health, we can focus on the nutrients required to produce and facilitate the uptake of thyroid hormones. The amino acid tyrosine is found in protein-rich foods such as almonds, pumpkin seeds, fish, chicken and also avocado. Iodine is found in seafood and seaweeds such as kelp, nori, dulse and arame, while selenium and zinc rich foods include oysters, tuna, chicken, mushrooms, Brazil nuts, almonds, cashews, oats, brown rice, buckwheat and quinoa.2 You should avoid refined carbohydrates, which affect blood sugar and have an impact on adrenal hormones, sex hormones and thyroid hormones, but should include plenty of protein and essential fats from oily fish, nuts, seeds, eggs, avocado and flaxseed to help stabilise hormones. If you have been diagnosed with an underactive thyroid, you should avoid eating large quantities of raw brassicas such as cabbage, kale and broccoli, but remember that lightly cooked green vegetables are a wonderful source of vitamins A and C, B vitamins and copper, which are needed for the thyroid gland to produce T4.9
In order to support your thyroid health, we can focus on the nutrients required to produce and facilitate the uptake of thyroid hormones. The amino acid tyrosine is found in protein-rich foods such as almonds, pumpkin seeds, fish, chicken and also avocado. Iodine is found in seafood and seaweeds such as kelp, nori, dulse and arame, while selenium and zinc rich foods include oysters, tuna, chicken, mushrooms, Brazil nuts, almonds, cashews, oats, brown rice, buckwheat and quinoa.2 You should avoid refined carbohydrates, which affect blood sugar and have an impact on adrenal hormones, sex hormones and thyroid hormones, but should include plenty of protein and essential fats from oily fish, nuts, seeds, eggs, avocado and flaxseed to help stabilise hormones. If you have been diagnosed with an underactive thyroid, you should avoid eating large quantities of raw brassicas such as cabbage, kale and broccoli, but remember that lightly cooked green vegetables are a wonderful source of vitamins A and C, B vitamins and copper, which are needed for the thyroid gland to produce T4.9

Stress management and regular exercise are equally as important for maintaining hormone balance, particularly adrenal hormones and in some cases, it is advisable to focus on improving adrenal health before supporting thyroid function. There is a clear correlation between food intolerances, gut permeability and auto-immune diseases, including auto-immune thyroid diseases such as Hashimoto’s.2 It is therefore crucial to support the gut by removing any foods to which the gut-based immune system is reacting and to heal the gut lining with nutrients such as l-glutamine.
In summary, the endocrine system is complex and should be considered in its entirety. An imbalance of adrenal hormones may have an impact on thyroid function and vice versa. Women with an underactive thyroid may find it harder to conceive and may struggle with the menopause. Whatever the cause, hypothyroidism can mean misery for many people if it remains undiagnosed, untreated, or mismanaged. It is important to conduct thorough testing through your GP, endocrinologist or nutrition practitioner and if medication is prescribed, to ensure that the dosage is regularly reviewed and adjusted, until you feel the benefits and see a reduction in symptoms. Certain other medications and nutritional supplements can interfere with the absorption and effectiveness of thyroid medication, so always check with your medical professional before taking anything new.
View List of References
- Gurel A, Dogantekin A, Ozkan Y, Aydin S (2015) Serum apelin levels in patients with thyroid dysfunction. International Journal of Clinical and Experimental Medicine, 8 (9): 16394-16398. [Online] PubMed Central (http://www.ncbi.nlm.nih.gov/pmc).
- Pizzorno J E, Murray M T, Joiner-Bey H (2008) The Clinicians Handbook of Natural Medicine, 2nd Edition. Churchill Livingstone, USA.
- Chakera A J, Pearce S H, Vaidya B (2012) Treatment for primary hypothyroidism: current approaches and future possibilities. Drug design, development and therapy, 6: 1-11. [Online] PubMed Central (http://www.ncbi.nlm.nih.gov/pmc)
- Malik R, Hodgson H (2002) The relationship between the thyroid gland and the liver. QJM, 95 (9): 559-569. [Online] QJM Oxford Journals (http://qjmed.oxfordjournals.org).
- Warner A, Mittag J (2012) Thyroid hormone and the central control of homeostasis. Journal of Molecular Endocrinology, 49: R29-R35. [Online] Journal of Molecular Endocrinology (http://jme.endocrinology-journals.org).
- Luongo C, Trivisano L, Alfano F, Salvatore D (2013) Type 3 deiodinase and consumptive hypothyroidism: a mechanism for a rare disease. Frontiers in Endocrinology, 4: 115. [Online] PubMed Central (http://www.ncbi.nlm.nih.gov/pmc).
- Roberts C G, Ladenson P W (2004) Hypothyroidism. The Lancet, 363 (9411): 793-803. [Online – abstract only] PubMed (http://www.ncbi.nlm.nih.gov/pubmed).
- Vanderpump M P J (2011) The epidemiology of thyroid disease. British Medical Bulletin, 99 (1): 39-51. [Online] British Medical Bulletin (http://bmb.oxfordjournals.org).
- Bajaj J K, Salwan P, Salwan S (2016) Various possible toxicants involved in thyroid dysfunction: a review. Journal of Clinical & Diagnostic Research, 10 (1): FE01-FE03. [Online] PubMed Central (http://www.ncbi.nlm.nih.gov/pmc).
- McNeil A, Stanford P (2015) Reporting thyroid function tests in pregnancy. The Clinical Biochemist Reviews, 36 (4): 109-126. [Online] Pub Med (http://www.ncbi.nlm.nih.gov/pubmed).
- Vanderpump M P J (2011) The epidemiology of thyroid disease. British Medical Bulletin, 99 (1): 39-51. [Online] British Medical Bulletin (http://bmb.oxfordjournals.org).
- Duffield A J, Thomson C D, Hill K E, Williams S (1999) An estimation of selenium requirements for New Zealanders. The American Journal of Clinical Nutrition, 70 (5): 896-903. [Online] The American Journal of Clinical Nutrition (http://ajcn.nutrition.org).
- Johannsson G, Skrtic S, Lennernas H, Quinkler M, Stewart P M (2014) Improving outcomes in patients with adrenal insufficiency: a review of current and future treatments. Current Medical Research and Opinion, 30 (9): 1833-1847. [Online – abstract only) PubMed (http://www.ncbi.nlm.nih.gov/pubmed).
- Saravanan P, Chau W F, Roberts N, Vedhara K, Greenwood R, Dayan C M (2002) Psychological well-being in patients on ‘adequate’ doses of l-thyroxine: results of a large, controlled community-based questionnaire study. Clinical Endocrinology, 57 (5): 577-585. [Online] PubMed (http://www.ncbi.nlm.nih.gov/pubmed).
- Surks M I, Boucai L (2010) Age- and race-based serum thyrotropin reference limits. The Journal of Clinical Endocrinology and Metabolism, 95 (2): 496-502. [Online] PubMed (http://www.ncbi.nlm.nih.gov/pubmed).
- Wartofsky L, Dickey R A (2005) The evidence for a narrower thyrotropin reference range is compelling. The Journal of Clinical Endocrinology and Metabolism, 90 (9): 5483-5488. [Online] PubMed (http://www.ncbi.nlm.nih.gov/pubmed).
- Pearce E N (2007) Thyroid dysfunction in perimenopausal and postmenopausal women. Post Reproductive Health, 13 (1): 8-13. [Online – abstract only] PubMed (http://www.ncbi.nlm.nih.gov/pubmed).