What should I eat?
‘Have you ever wondered why some people can eat a high fat diet and have no problem with cholesterol, or why others can have an after-dinner espresso and not be kept awake all night?’
Nutrigenomics is an emerging science which investigates how the foods we eat interact with our genes, to affect our health. The human genome project was completed in 2003 and enabled us for the first time, to read nature’s complete genetic blueprint for building a human being. As humans, we are 99.9% genetically identical to each other, but we vary slightly due to variations in certain genes.1,2 These are sometimes called gene mutations or SNPs (pronounced as ‘snips’ and an abbreviation for single nucleotide polymorphism, meaning that a tiny part of the DNA pattern differs from one variation of a gene to another). Have you ever wondered why some people can eat a high fat diet and have no problem with cholesterol, or why others can have an after-dinner espresso and not be kept awake all night? Due to these common variations in genes, we are beginning to understand how individuals respond differently to dietary intake, medication, or lifestyle factors such as stress, toxic exposure and exercise.

How can our genes determine our responses to the food we eat?
The goal of the human genome project was to provide researchers with powerful tools to understand the genetic factors in human disease. The field of nutrigenomics is showing us how nutrients and bioactive components in food can ‘switch on or off’ certain genes, which then impact on important metabolic and physiological processes in the body, such as detoxification and energy production. A significant example is sulforaphane, a compound produced through digestion of cruciferous vegetables, particularly broccoli and broccoli sprouts. Sulforaphane upregulates, or ‘switches on’ a variety of genes that give the body instructions to make proteins which protect cells from damage: in other words, it contributes to switching on our internal defence and ‘spring cleaning’ mechanisms. In this way, sulforaphane has been shown to be involved in inhibiting inflammation, cancer development and the build-up of toxins which can damage cells and lead to disease development. 3-5
How do we send messages to our cells?
Each of o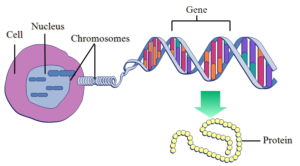 ur ≃21,000 genes6 provide messages that instruct your body to make a specific protein (these proteins are involved in creating and maintaining your functioning body). When genes switch on and send a message to cells, it is called ‘gene expression.’ For example, when we digest carbohydrate such as rice, it is converted to glucose and absorbed into our bloodstream. The INS gene is expressed, or ‘switched on’ and sends a coded message to the pancreas, telling it to produce a certain amount of the protein hormone insulin. Insulin directs the glucose out of the blood and into body cells, to be turned into energy. Once blood glucose levels return to normal, the INS gene is switched off and the pancreas stops producing insulin. Mutations in this gene cause an error in communication with the pancreas and have been linked to diabetes in newborns.7
ur ≃21,000 genes6 provide messages that instruct your body to make a specific protein (these proteins are involved in creating and maintaining your functioning body). When genes switch on and send a message to cells, it is called ‘gene expression.’ For example, when we digest carbohydrate such as rice, it is converted to glucose and absorbed into our bloodstream. The INS gene is expressed, or ‘switched on’ and sends a coded message to the pancreas, telling it to produce a certain amount of the protein hormone insulin. Insulin directs the glucose out of the blood and into body cells, to be turned into energy. Once blood glucose levels return to normal, the INS gene is switched off and the pancreas stops producing insulin. Mutations in this gene cause an error in communication with the pancreas and have been linked to diabetes in newborns.7
‘…we need to be cautious of media hype, whereby one day coffee is ‘good’ for us and another day ‘bad,’ because the way that each individual responds to something like caffeine can vary so drastically…’
Certain genes can affect the rate that we break down, use and get rid of almost everything we consume. Taking caffeine as an example, the gene which instructs how caffeine is broken down in the liver is CYP1A2. Individuals with what is referred to as a ‘fast’ version of the CYP1A2 gene can metabolise (break down) and excrete caffeine quickly, while those with a slower version cannot.8 It is also interesting that the gene is more active in men than women and that it is inhibited by taking the contraceptive pill.9 This type of information becomes more useful when used as part of larger studies, such as those which show that fast metabolisers of caffeine have a lower risk of heart attack when increasing consumption of caffeinated coffee. This may have nothing to do with the caffeine, but could be that having quickly removed the caffeine from their body, they are able to make better use of the heart-protective compounds in the coffee. 10 Other studies have shown that slow metabolisers of caffeine are more likely to experience high blood pressure, as the caffeine has a prolonged stimulatory effect on their adrenal system. 11 This is where we need to be cautious of media hype, whereby one day coffee is ‘good’ for us and another day ‘bad,’ because the way that each individual responds to something like caffeine can vary so drastically and can be linked back to their individual genetic profile. Indeed, where many studies show conflicting results, it is worth bearing in mind that they might not have accounted for genetic influence.
How can I apply nutrigenomics to my everyday diet?
Fish oil is a much-confused subject, which receives frequent media attention. A few studies have now shown that people who have a particular version of the PPARϒ gene respond much better to the blood lipid-lowering effect of fish oils (polyunsaturated fats), with subsequent reduction in risk of heart disease.12 Individuals with other variations of the PPARϒ gene fare better when monounsaturated fats (e.g. olive oil, coconut oil) are consumed in greater quantities.13 We also know that zinc is a vital component of PPARϒ gene expression14 and that alcohol inhibits the gene, so by understanding which version of the gene we inherited, we are able to take a proactive approach to managing the risk of weight gain, type 2 diabetes and alcohol-associated breast cancer, which are linked with certain variations of the PPARϒ gene. For example, we can adjust the ratios of the types of fats we eat, understanding that while oily fish, avocado and sunflower seeds are all generally accepted to be healthy foods, that more of one and less of another may be more beneficial to us as an individual, depending on which variation of the PPARϒ gene we have. We can also monitor our zinc levels, adjusting diet or supplements in a more targeted way and can make more informed decisions about our alcohol intake.



Enabling a more personalised supplementation programme
…the methylation process is critical to overall health…’
Folic acid is a synthetic (chemically man-made) form of vitamin B9, which is used in many supplements and fortified foods including wheat flour and breakfast cereals, while folate is the natural form of vitamin B9, found in green leafy vegetables such as broccoli and spinach. Pregnant women are recommended to supplement 400mcg folic acid daily in their first trimester, to prevent birth defects, however we now know that for some women with a very common genetic mutation, this may not be as beneficial as they would hope. 15,16 Both synthetic folic acid and natural folate need to be converted by the body, through various stages, into an active form of the vitamin B9. This process requires the MTHFR gene to produce an enzyme called methylenetetrahydrofolate reductase (hence its abbreviation to MTHFR!). It is estimated that 60% of the US population have a mutated variant of this gene, which results in the enzyme activity being less effective i.e. they are less capable of converting folic acid and folate to its active form. This has a knock-on effect on many processes and has been linked to various health issues, including cardiovascular disease; recurrent miscarriage; migraine; mental disorders such as autism, anxiety or depression; and digestive problems such as IBS.
 It is important to note that not everyone with the MTHFR mutation will experience associated health issues, but the methylation process is so critical to overall health, that it is very useful to know which variant of the gene you were born with. Those with mutated variants can be guided towards making better choices of activated B vitamin supplements and to keeping a closer eye on certain blood markers such as vitamin B12, cholesterol and homocysteine, which are affected by inefficient conversion of dietary folate or supplemental folic acid to its active form.
It is important to note that not everyone with the MTHFR mutation will experience associated health issues, but the methylation process is so critical to overall health, that it is very useful to know which variant of the gene you were born with. Those with mutated variants can be guided towards making better choices of activated B vitamin supplements and to keeping a closer eye on certain blood markers such as vitamin B12, cholesterol and homocysteine, which are affected by inefficient conversion of dietary folate or supplemental folic acid to its active form.
Supporting a targeted exercise and weight management regime
‘…some genetic profiles respond better to aerobic exercise and are able to burn fat efficiently, while others are so predisposed to inflammation, that they may achieve much better results with less intense exercise…’
Do you spend hours at the gym, but can’t seem to shift weight, or do you experience delayed onset muscle soreness after exercise? The reasons may lie in your genes and by knowing which of your genes influence inflammation and fat storage, for example, you can make better choices about the way you exercise. Studies show that some genetic profiles respond better to aerobic exercise and are able to burn fat efficiently,17 while others are so predisposed to inflammation, that they may achieve much better results with less intense exercise, focusing on resistance training,18 combined with active recovery sessions, such as gentle walking or cycling. For some people, seeing their genetic profile can be a light bulb moment, as they gain a clearer picture of some of the reasons behind their difficulty to lose weight or regulate appetite and understand their risk of obesity (including childhood obesity) or diabetes. When this information is presented alongside the scientific research to support which measures can be taken to influence specific genes, it provides actionable areas to target, in terms of diet, lifestyle supplements and exercise.
Knowledge is power
It is important to understand that we cannot change the genes that we were born with, however, we can influence the way that they express themselves (switch on and off) and impact on our health. Good health is determined by healthy cells. We constantly replace most of the cells in our body and can choose to optimise cell function by sending messages through the food we eat and lifestyle choices that we make, such as smoking, stress management and physical exercise.
A genetic test and in-depth analysis can help you to understand which foods will benefit you the most, which nutritional supplements could enhance your wellbeing and which type of exercise will have a positive effect on your health. Rather than trying to work out which of the latest scientific research is relevant to you, this can take out some of the guesswork and enable you to optimise your health with a truly personalised plan. If you eat healthily and exercise regularly, but cannot shift weight, understanding your genetic profile could help to target your diet and exercise regime. If you have a history of heart disease, cancer, diabetes or other major diseases in your family and would like to better understand your risk and then take a more-informed, proactive approach to your health, this is a test for you.
On a personal note
Having completed a FitGenes profile myself, I am now aware of the health issues to which I am more predisposed. While I currently enjoy excellent health, I feel empowered knowing which of those potential health issues to focus on and I have already made changes to what I previously believed to be extremely healthy diet and exercise patterns. I have always known that lack of sleep through my twenties (work hard, play hard decade) and thirties (kittens and babies, plus work hard and much less play hard decade) was in no way beneficial to my health, I now appreciate just what a priority I need to make sleep as I start my forties decade! I have made further tweaks to my diet, changed the supplements I take and altered my exercise routine. Rather than taking my current state of good health for granted, I am taking proactive steps to monitor and manage my areas of risk, for example, undertaking further diagnostic tests such as a complete hormone profile and keeping a closer eye on markers of inflammation. My biggest challenge is to improve the quality of my sleep, which like so many people, is all too often pushed down the priority list, in an attempt to gain more hours in the day.
I would encourage you to take a proactive approach to your health – whether you are hoping to conceive a child (yes, you too dads-to-be, it takes two sets of genes to make a baby); determined to optimise your exercise training; attempting to reduce or manage risk of family diseases; or simply wanting to age healthily. Please see here for further details of what a FitGenes test involves and contact me if you have any questions. To quote Dr Paul Beaver, co- founder of FitGenes, ‘your genes are not your destiny!’
View List of References
-
- National Human Genome Research Institute Presentation (2008) Human Genetic Variation. [Online] National Human Genome Research Institute (www.genome.gov/pages/education/modules/geneticvariation.pdf)
- Bethedsa (2007) Understanding Human Genetic Variation. National Institutes of Health (US) Curriculum Supplement Series. [Online] National Center for Biotechnology Information (www.ncbi.nlm.nih.gov).
- Tortorella S M, Royce S G, Licciardi P V, Karagiannis T C (2015) Dietary Sulforaphane in Cancer Chemoprevention: The Role of Epigenetic Regulation and HDAC Inhibition. Antioxidants and Redox Signalling, 22 (16): 1382-1424. [Online] PubMed Central (www.ncbi.nlm.nih.gov/pmc).
- Guerrero-Beltran C E, Calderon-Oliver M, Pedraza-Chaverri J, Chirino Y I (2012) Protective effect of sulforaphane against oxidative stress: recent advances. Experimental and Toxicologic Pathology, 64 (5): 503-508. [Online] PubMed (www.ncbi.nlm.nih.gov/pubmed).
- Yang L, Palliyaguru D L, Kensler T W (2016) Frugal Chemoprevention: Targeting Nrf2 with Foods Rich in Sulforaphane. Seminars in Oncology, 43 (1): 146-153. [Online] Seminars in Oncology (www.seminoncol.org).
- Frazer K A (2012) Decoding the human genome. Genome Research, 22: 1599-1601. [Online] Genome Research (www. genome.cshlp.org).
- Edghill E L et al. (2008) Mutations in the INS Gene Are a Common Cause of Neonatal Diabetes but a Rare Cause of Diabetes Diagnosed in Childhood or Adulthood. Diabetes Journals, 57 (4): 1034-1042. [Online] Diabetes Journals (www. diabetes.diabetesjournals.org).
- Cornelis M C, El-Sohemy A, Kabagambe E K, Campos A H (2006) Coffee, CYP1A2 Genotype, and Risk of Myocardial Infarction. Journal of the American Medical Association, 295(10): 1135-1141. [Online] The JAMA Network (www.jamanetwork.com).
- Rasmussen B B, Brix T H, Kyvik K O, Brosen K (2002) The interindividual differences in the 3-demthylation of caffeine alias CYP1A2 is determined by both genetic and environmental factors. Pharmacogenetics, 12 (6): 473-478. [Online] Pharmacogenetics and Genomics (www.journals.lww.com/jpharmacogenetics).
- Butt M S & Sultan M T (2011) Coffee and its Consumption: Benefits and Risks. Critical Reviews In Food Science And Nutrition, 51 (4). [Online] Taylor & Francis Online (www.tandfonline.com).
- Palatini P, Ceolotto G, Ragazzo F, Dorigatti F, Saladini F, Papparella I, Mos L, Zanata G, Santonastaso M (2009) CYP1A2 genotype modifies the association between coffee intake and the risk of hypertension. Journal of Hypertension, 27 (8): 1594-1601. [Online] Journal of Hypertension (www.journals.lww.com/jhypertension).
- Lindi V, Schwab U, Louheranta A, Laakso M, Vessby B, Hermansen K, Storlien L, Riccardi G, Rivellese A (2003) Impact of the Pro12Ala polymorphism of the PPAR-gamma2 gene on serum triacylglycerol response to n-3 fatty acid supplementation. Molecular Genetics & Metabolism, 79 (1): 52-60. [Online] PubMed (www.ncbi.nlm.nih.gov).
- Soriguer F, Morcillo F, Cardona F, Rojo-Martinez G, Almaraz M, Ruiz de Adana M, Olveira G, Tinahones F, Esteva I (2006) The Journal of Nutrition, 136 (9): 2325-2330. [Online] The Journal of Nutrition (www. jn.nutrition.org).
- Meerarani P, Reiterer G, Toborek M, Hennig B (2003) Zinc modulates PPARgamma signaling and activation of porcine endothelial cells. Journal of Nutrition 133 (10): 3058-64. [Online] PubMed (www.ncbi.nlm.nih.gov).
- Greenberg J A, Bell S J, Guan Y, Yu Y-H (2011) Folic Acid Supplementation and Pregnancy: More Than Just Neural Tube Defect Prevention. Reviews in Obstetrics and Gynaecology, 4 (2): 52-59. [Online] PubMed Central (www.ncbi.nlm.nih.gov/pmc).
- Scholl T O & Johnson W G (2000) Folic acid: influence on the outcome of pregnancy. American Journal of Clinical Nutrition, 71 (5): 1295s-1303s. [Online] American Journal of Clinical Nutrition (ajcn.nutrition.org).
- Macho-Azcarate T, Calabuig J, Marti A, Martinez J A (2002) A maximal effort trial in obese women carrying the β2-adrenoceptor Gln27Glu polymorphism. Journal of Physiology and Biochemistry, 58 (2): 103-108. [Online] SpringerLink (www.link.springer.com).
- Yao, L, Delmonico M J, Roth S M, Hand B D, Johns J, Conway J, Douglass L, Hurley B F (2007) Adrenergic Receptor Genotype Influence on Midthigh Intermuscular Fat Response to Strength Training in Middle-Aged and Older Adults. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 62 (6): 658-663. [Online] PubMed Central (www.ncbi.nlm.nih.gov/pmc).